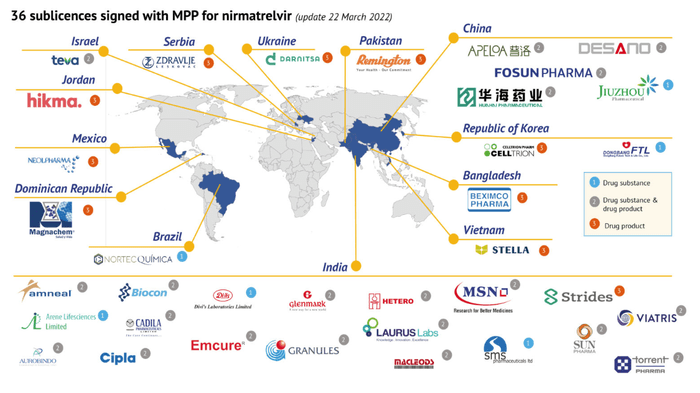
Caption: Through Medicine Patent Pool, a WHO backed global health agency, Pfizer has granted sub-licenses for36 pharmaceutical manufacturers around the globe to make generic versions of Paxlovid. -Source http://www.MedicinePatentPool.com
Because the skyrocketing number of COVID cases leads to severe shortage of effective anti-COVID-viral agents in China, the life-saving drug, Paxlovid, now is sold in unofficial channels at a price as much as more than 50,000 RMB for one package ( one course treatment), reportedly. On January 8, the National Health Security Administration said that insurance coverage of Paxlovid would be ended before April due to the failure of price negotiation with the manufacturer of Paxlovid, Pfizer. This unfortunate development will probably fan people’s panic in procuring the drug. E-commerce platforms of selling the so called generic versions of Paxlovid, as well as some online media are seemingly celebrating this golden opportunity, starting all engines to promote their products. Articles cheering for those generics have an eye-popping headlines such as “ Indian generics emerges as a life-saver in COVID hit China” (offonline.org), “Demand for Indian generic drugs skyrockets in China amid massive Covid surge” (–economictimes.indiatimes.com). On another hand, voices have ranked out to alert the public that the counterfeits of Paxlovid as well as so called generic versions of Paxlovid are circulating over e-commerce platforms. One article, in particular, published on January 6 in a Shanghai based media website, sixthtone.com, has reported details of an ugly picture of fake generics of paxlovid. In the present situation, with absence of official surveilling mechanism, the black market is thriving. People are left bewildering in making decision whether to pay a loft price for the urgently needed medication, “Is this anti-COVID-viral, supposed to be a live-saving drug, made of potato starch?”
Primovir is labeled as a generic version of Paxlovid, made by Astrica, an Indian company. According to the report from Sixthtone, buyers of Primovir from online source have been sending their purchased Primovir for lab tests. Paxlovid package contains two different tablets, one is nirmatrelvir and another is ritonavir. One buyer who was able to access the testing technique found out that the nirmatrelvir tablet in the Primovir package did not contain the main active anti-COVID-viral ingredient, nirmatrelvir. After this initial finding, a Chinese genomic company called BGI Group has started testing those so called generics of Paxlovid, for the public, free of charge. The lab of BGI has tested 150 package samples to the date of January 6. Among 143 package samples of Primovir, only one has nirmatrelvir detected. Products of some other brands were found indeed containing nirmatrelvir, BGI reported.
Paxista is another one labeled as generic version of Paxlovid, made by Azista Bhutan Health Care, also an Indian company. In a BGI’s report, the results of tests showed that nirmatrelvir was detected in Paxista. However, this singularly positive result is probably still thousand miles away from anything to support the claim that Paxista is a generic sub-licensed by Pfizer. Through Medicine Patent Pool, a WHO backed global health agency, Pfizer has granted sub-licenses for36 pharmaceutical manufacturers around the globe to make generic versions of Paxlovid. The maker of Paxista, Azista, is not listed among the 35 manufacturers. This is further confirmed by my communication with Pfizer. My inquiry sent to Azista received no response. Hetero Health Care is Azista’s sister company, and in the list of sub-licences, but Hetero is still in a process of developing its own generic for Paxlovid, under the name “ Nirmacom”. On Dec 27 2022, Hetero Health Care stated that Hetero’s ‘Nirmacom’ is world’s first generic version of COVID-19 oral drug ‘PAXLOVID’ to receive WHO Prequalification (quoted from prnewwire.com, Ciaion Communication). It is intriguing, but it would be reasonable to believe that Hetero would not be silly enough to tarnish its reputation by launching its product prior to approval in black market through Azista.
There are myriad of unanswered questions about those drugs made in India, labeled as generics of Paxlovid. One of poignant questions is why those drugs have the caution note in their package “Not for domestic use, for export only”? Does it stamp the caution note on its products in order to dodge the surveillance of Indian administration, and then be free of constraint to influx into other countries like China? A status of generic drugs is strictly described as pharmaceutically equivalent or therapeutically equivalent. To evaluate a generic drug, bioavailability is one of the most important criteria, which is the measurement of levels of a drug absorbed into the body system. This comparison between a original brand drug and its generic version enables the evaluation of a generic regarding its potency and efficacy. The positive result of detecting nirmatrelvir in Paxista is merely enough to give patients any relief of a trust.
Five Chinese pharmaceutical companies have signed the agreement with Medicine Patient Pool for the sub-licenses of manufacturing generics of Paxlovid. On January 9, the CEO of Pfizer said that a Chinese pharmaceutical company, supposedly Zhejiang Huahai Pharmaceutical, would start the production of brand Paxlovid in the first half of the year 2023. It seems until then, the shortage of life-saving Paxlovid would continue.
Paxlovid is so difficult to access, how about other anti-COVID-virals ?
Molnupiravir is another oral anti-COVID- viral medicine approved by FDA in December 2021. It reduces the risks of hospitalization and death by 30%. It is recommended for use only when no other more effective treatment is available. The original manufacturer, Merck, signed an agreement with MPP in October 2021, authorized MPP sub-licensed 25 countries for the production of its generic version. China is not in the list of 25 countries, but several Chinese pharmaceutical companies have the partnership with Merck for the generic production of molnupiravir.
Remdesivir is a intravenous injectable anti-COVID-viral medicine. It shortens the time course of hospitalization for COVID treatment by 33%. Since it was approved by FDA in May 2020, its indication has been expanded for both hospitalized and non-hospitalized patients, as well as age groups. At present, it is commonly used in hospitals in US. Remdesivir has not gained the approval of use in China. But Chinese scientists are in a process of developing an oral dosage form of remdesivir.
All in all, there is no an immediate solution for the urgent need of anti-COVID-virals. When people are pondering and turning their sight towards the black markets for so called “generics”, what better advice could be given? Although verification of claims of many online published reports is necessary, including that report of disclosing the fake Paxlovig, the answer is always the same, “careful”.
It has been of a great encouragement and proud to see that many oversea Chinese physicians in U.S. voluntarily organized themselves in a conference in New Year Day of 2023, to share their clinical experiences with the physicians in China to save COVID patients’ lives. With the organization, a scientist of solid pharmaceutical research background spares no effects in combating various toxic misguiding rumors. Online circulating rumors, which are like poisonous weeds, have been so harmful to innocent people’s health, particularly in the understandings of COVID vaccination and treatment. I and many others have to salute those oversea-Chinese physicians carrying on their benevolent cause. For the pharmaceutical manufacturers as well as the black market vendors of labeled “generic versions” of Paxlovid, let us bear a message in mind “ For what it profit a man, if he shall gain the whole world, and lose his own soul” (Mark 8 – 36)
Ref:
In China, a Dark New Black Market Emerges: Fake COVID Medication, Wu Peiyue, Sixthtone.com, Jan 6 2023
Update: Ukrainian company Darnitsa signs sublicence agreement with MPP bringing to 36 the number of generic manufacturers to produce generic versions of Pfizer’s oral COVID-19 treatment. MedicinePatentPool.com/News, 22 March 2022
The Medicines Patent Pool (MPP) and MSD enter into licence agreement for molnupiravir, an investigational oral antiviral COVID-19 medicine, to increase broad access in low- and middle- income countries, MedicinePatentPool.com/news, 27 October 2021
FOR PATIENTS HOSPITALIZED WITH COVID-19,HELP REDUCE DISEASE PROGRESSION AND SHORTEN RECOVERY TIME, 2022 Gilead Sciences, Inc. US-VKYP-0178 12/22,Vekluryhcp.com
VV116 versus Nirmatrelvir–Ritonavir for Oral Treatment of Covid-19, Zhujun Cao at al, New England Journal of Medicine, December 28; DOI: 10.1056/NEJMoa2208822